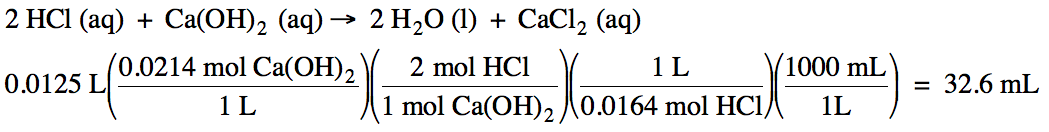Chapter 10: Chemical Tests and Chemical Reaction
Types
Section 10-1: Laboratory Tests to Identify Chemicals
Section 10-2: Diatomic Nonmetals
Section 10-3: Combustion of Organic Compounds
Section 10-4: Reactions Involving Metals or Metal Compounds
Section 10-5: Aqueous Ionic Compounds and Molarity of Ions
Section 10-6: Solubility Rules, Precipitation Reactions, and Net Ionic Equations
Section 10-7: Acid + Base Neutralization Reactions and Molecular Equations
Section 10-8: Experiment - Acid + Base Titration
Chapter 10 Practice Exercises and Review Quizzes
Section 10-1: Laboratory Tests to Identify Chemicals
The following lab tests have been
used to indicate the likely presence of the common chemicals listed:
|
Chemical |
Test |
|
carbon dioxide gas, CO2 (g) |
flaming wooden splint extinguished |
|
hydrogen gas, H2 (g) |
flaming wooden splint "pops"
|
|
oxygen gas, O2 (g) |
glowing wooden splint relights |
|
significant concentration of aqueous
hydrogen ion, H+
(aq) |
solution is acidic, so: 1. low pH meter reading 2. litmus paper will be red 3. phenolphthalein indicator will be colorless |
|
significant
concentration of aqueous hydroxide ion,
OH- (aq) |
solution is basic, so: 1. high pH meter reading 2. litmus paper will be blue 3. phenolphthalein indicator will be pink |
Section 10-2: Diatomic Nonmetals
There are 7 nonmetals that exist in
covalently-bonded pairs at room temperature and pressure. Each of these is listed below along
with its state of matter at room temperature and pressure:
hydrogen gas, H2 (g)
nitrogen gas, N2 (g)
fluorine gas, F2 (g)
chlorine gas, Cl2 (g)
liquid bromine, Br2 (l)
solid iodine, I2 (s)
When the elements above are
reactants or products in a chemical reaction, they should be written as
diatomic.
Section 10-3: Combustion of Organic Compounds
The complete combustion or burning
of organic compounds with the condensed formula CwHxOySz requires sufficient oxygen gas and will generally produce carbon dioxide, water, and sulfur dioxide:
CwHxOySz + O2 → CO2 + H2O + SO2
The products will be the same regardless of whether y = 0 or y > 0 or, in other
words, regardless of whether or not the compound burned contains oxygen. If the compound burned does not contain carbon (w = 0), then no carbon dioxide will be produced. If the compound burned does not contain hydrogen (x = 0), then no water will be produced. If the compound burned does not contain sulfur (z = 0), then no sulfur dioxide will be produced:
Sample Exercise 10A:
Write a balanced equation for the
combustion of each compound below using the smallest possible whole-number
coefficients:
(a) hexane
(b) propanol
(c) carbon disulfide
Solution:
(a) 2 C6H14 +
19 O2 → 12 CO2
+ 14 H2O
(b) 2 C3H7OH
+ 9 O2 → 6 CO2
+ 8 H2O
(c) CS2 + 3 O2→ CO2 + 2 SO2
Section 10-4: Reactions Involving Metals or Metal
Compounds
Neutral metals or compounds
containing metal ions are reactants in the following reactions:
(a) metal + nonmetal → ionic compound (also known as ionic salt)
For example, lithium metal reacts
with nitrogen gas to produce lithium nitride according to the following
balanced equation:
(lithium nitride = combine 3 Li+ and N3-
= Li3N)
6 Li + N2
→ 2 Li3N
(b) metal + water → hydrogen gas + metal cation
+ hydroxide ion
For example, sodium metal reacts
with water according to the following balanced equation:
(be sure that the metal cation has
the correct charge)
2 Na + 2 H2O
→ H2
+ 2 Na+ + 2 OH-
(note that the coefficient 2 in front of the Na+ is added to ensure that the total positive charge equals the total negative charge on the right side of the equation)
(c) metal oxide + water → metal cation + hydroxide
ion
For example, potassium oxide reacts
with water as follows:
(potassium oxide = combine 2 K+ and O2-
= K2O)
K2O
+ H2O → 2 K+
+ 2 OH-
(d) metal carbonate → metal oxide + carbon dioxide
For example, magnesium carbonate
decomposes when heated to produce magnesium oxide and carbon dioxide according
to the following balanced equation:
(magnesium carbonate = combine Mg2+ and CO32-
= MgCO3,
magnesium oxide = combine Mg2+ and O2- = MgO)
MgCO3
→ MgO + CO2
Sample Exercise 10B:
Write a balanced equation for each
reaction described below using the smallest possible whole-number coefficients:
(a) aluminum
metal reacts with solid iodine
(b) calcium
metal reacts with water
(c) strontium
oxide reacts with water
(d) sodium
carbonate decomposes upon heating
Solution:
(a) (product
is aluminum iodide = combine Al3+ and 3 I- = AlI3)
2 Al + 3 I2
→ 2 AlI3
(b) (metal
cation produced is Ca2+)
Ca + 2 H2O
→ H2
+ Ca2+ + 2 OH-
(c) (strontium
oxide = combine Sr2+ and O2- = SrO)
SrO + H2O → Sr2+
+ 2 OH-
(d) (sodium
carbonate = combine 2 Na+ and CO32- = Na2CO3,
product is sodium
oxide = combine 2 Na+ and O2- = Na2O)
Na2CO3
→ Na2O
+ CO2
Section 10-5: Aqueous Ionic Compounds and Molarity of Ions
When a solid ionic compound is
dissolved in water, the ions separate from each other and become surrounded by
water molecules in the aqueous solution.
For example, KI separates in water into one K+ cation and one I- anion per formula unit, while
(NH4)2SO4 separates
in water into two NH4+ cations
and one SO42- anion per formula unit. A molarity
written on the bottle of an aqueous ionic compound will be accompanied by the
formula of the neutral ionic compound but will not specify the molarity of individual ions in the solution. As such, if we wish to know the molarity of each type of ion in the solution, we must
translate the information on the bottle using the number of cations
and anions per formula unit of the ionic compound. For example, in a bottle labeled 0.026 M Na3PO4,
the molarity of Na+ will be 3 x 0.026 M =
0.078 M Na+ because there are 3 Na+ cations
per formula unit. The molarity of PO43- will be 1 x 0.026 M
= 0.026 M PO43- since there is one PO43-
anion per formula unit.
Sample Exercise 10C:
What is the molarity
of each ion in the following solutions?
(a) 1.2 M CaCl2
(b) 0.31 M Al(NO3)3
Solution:
(a) 1 x 1.2 M = 1.2 M Ca2+
and 2 x 1.2 M = 2.4 M Cl-
(b) 1 x 0.31 M = 0.31 M Al3+
and 3 x 0.31 M = 0.93 M NO3-
Section 10-6: Solubility Rules, Precipitation
Reactions, and Net Ionic Equations
A solid ionic compound that
dissolves significantly in water is said to be soluble. A solid ionic compound that does not
dissolve to a significant extent in water is said to be insoluble. We will use the following widely accepted general
solubility rules to determine which common ionic compounds will be soluble
versus insoluble in water (solubility rules from other sources may differ
slightly):
Water Solubility of Ionic Compounds
1) always soluble: compounds containing Group 1 alkali
metal cations, NH4+, CH3COO-, or NO3-
2) usually soluble: compounds containing Br-, Cl-,
or I- (except when combined with Ag+, Hg22+,
or Pb2+)
3) usually soluble: compounds containing SO42- (except when combined with Ba2+, Ca2+, Pb2+,
or Sr2+)
4) usually insoluble: compounds containing CO32-,
CrO42- (chromate), or PO43- (except
when combined with Group 1 alkali metal cations or NH4+)
5) usually insoluble: compounds containing OH- or
S2- (except when combined with Group 1 alkali metal cations, NH4+,
Ba2+, Ca2+, or Sr2+)
When two different ionic compound
solutions are mixed, a reaction will occur if cations from one solution can
combine with anions from the other solution to form a solid precipitate that is insoluble. When the solutions Mg(NO3)2 (aq) and K3PO4 (aq) are mixed, we can eliminate one
combination of cation + anion that cannot possibly form an insoluble solid
precipitate in water according to the solubility rules above, and we can also
determine that the second combination of cation + anion will form an insoluble
precipitate:
K+ (aq) (Group 1 alkali metal cation) + NO3- (aq) → no reaction [Rule 1]
Mg2+ (aq) + PO43- (aq) → Mg3(PO4)2 (s) [Rule 4]
The balanced equation for the
reaction of magnesium cations and phosphate anions is known as a net ionic equation because it only
includes the ions that are truly involved in the precipitation reaction:
net ionic
equation: 3 Mg2+ (aq) +
2 PO43- (aq) → Mg3(PO4)2 (s)
The K+ cations and NO3- anions are known as spectator ions because, although they are present in the solution, they do not actually take
part in the reaction and, thus, are omitted from the net ionic equation.
If neither combination of cation +
anion can form an insoluble solid precipitate, all ions will continue to exist
separately in the mixture of solutions and, thus, no overall reaction
occurs. For example, when a
solution of lithium sulfate is mixed with a solution of ammonium nitrate, neither
combination of cation from one solution + anion from the second solution can
form an insoluble solid precipitate, so no overall reaction occurs and no solid
will be observed:
Li+ (aq) + NO3- (aq) → no
reaction
NH4+ (aq) + SO42- (aq) → no
reaction
Sample Exercise 10D:
Indicate if any of the combinations
below yield no reaction and also write a balanced net ionic equation, including
states of matter, for any combinations that do yield a precipitate:
(i) AgNO3 (aq) + (NH4)2CO3 (aq)
(ii) NaNO3 (aq) + CuSO4 (aq)
(iii) aqueous ammonium chloride +
aqueous lithium nitrate
(iv) aqueous potassium hydroxide +
aqueous nickel(II) nitrate
Solution:
No
reaction will occur for (ii) and (iii) as all combinations of cation + anion
will be soluble. One precipitate
will form in each of (i) and (iv):
(i)
NH4+ (aq) + NO3- (aq) → no reaction
net ionic equation: 2 Ag+ (aq) + CO32- (aq) → Ag2CO3 (s)
(ii) Na+ (aq) + SO42- (aq) → no
reaction
Cu2+ (aq) + NO3- (aq) → no
reaction
(iii) NH4+ (aq) + NO3- (aq) → no reaction
Li+ (aq) + Cl- (aq) → no reaction
(iv) K+ (aq) + NO3- (aq) → no
reaction
net ionic equation: Ni2+ (aq) + 2 OH- (aq) → Ni(OH)2 (s)
Note that it is also possible for both combinations of cation + anion to yield a precipitate, in
which case a mixture of two different solid precipitates will be produced in
the same reaction. For example,
when CoSO4 (aq) is mixed with Ba(OH)2 (aq), two
precipitates are formed simultaneously according to the following net ionic
equation:
Co2+ (aq) + 2 OH- (aq) + Ba2+ (aq) + SO42- (aq) → Co(OH)2 (s) + BaSO4 (s)
Section
10-7: Acid + Base Neutralization
Reactions and Molecular Equations
The
reaction of an aqueous acid with an aqueous metal hydroxide can be considered a
neutralization reaction because the acidic H+ ions from the acid
solution and the basic OH- ions from the metal hydroxide solution
combine to form neutral water.
Although these reactions can be written as net ionic equations, we can
also write a molecular equation with
spectator ions included as follows:
acid (aq) +
metal hydroxide (aq) → water (l) + ionic compound (or ionic salt) (aq)
The
correct formula of the ionic compound (or ionic salt) produced is a combination
of the metal cation from the metal hydroxide and the anion from the acid. For example, in the reaction of
sulfuric acid with sodium hydroxide, the Na+ cations from the NaOH
combine with the SO42- anions from the H2SO4
to yield the formula Na2SO4 in the balanced molecular
equation:
H2SO4 (aq) + 2 NaOH (aq) → 2 H2O (l) + Na2SO4
(aq)
Sample
Exercise 10E:
Write
a balanced molecular equation, including states of matter, for the
neutralization reaction between solutions of acetic acid and calcium hydroxide.
Solution:
For
neutralization reactions, we will write acetic acid as HCH3COO:
2 HCH3COO (aq) + Ca(OH)2 (aq) → 2 H2O (l) + Ca(CH3COO)2
(aq)
Note
that it is possible for the ionic compound produced to be an
insoluble solid rather than aqueous based on the solubility rules above. For example, when solutions of sulfuric
acid and barium hydroxide are mixed, the balanced molecular equation is:
H2SO4 (aq) + Ba(OH)2 (aq) → 2 H2O
(l) + BaSO4 (s)
Section
10-8: Experiment – Acid +
Base Titration
In a
titration experiment, a precisely
measured volume of one reactant solution is added dropwise from a buret (or
burette) to a second reactant solution until the stoichiometric equivalence
point of the reaction has been reached.
When the stoichiometric equivalence point has been reached, neither
reactant is in excess and, therefore, we can use the mole ratio from the
balanced chemical reaction in stoichiometry calculations to determine the
unknown molarity of one of the reactants.
One
common titration experiment utilizes an aqueous acid + aqueous metal hydroxide
neutralization reaction. An acid
of known molarity and volume is placed in an Erlenmeyer flask along with a few
drops of phenophthalein indicator, which will be colorless in the acid
solution. A metal hydroxide
solution with unknown molarity is then added dropwise from a buret until the
phenolphthalein indicator in the mixture obtains a faint pink color signifying
that the endpoint has been reached.
Although this pink endpoint is caused by a very slight excess of basic
hydroxide ions from the final drop of metal hydroxide solution that was added,
we can assume that the exact stoichiometric equivalence point has been reached
and, therefore, that neither the acid nor the metal hydroxide is in excess at
that point.
After
calculating the known moles of the acid, we can use the mole ratio from the
balanced molecular equation to calculate the unknown moles of the metal
hydroxide added from the buret. We
then divide by the volume in liters of the metal hydroxide solution added from
the buret to obtain the unknown molarity of the metal hydroxide solution. For example, if we start with 16.7 mL
of 0.116 M hydrochloric acid in the Erlenmeyer flask and 14.9 mL of sodium
hydroxide solution is required to titrate the acid (reach the equivalence
point), the unknown molarity of the sodium hydroxide solution can be determined
as follows:

Note
that the calculated molarity of the NaOH solution may differ from the true
value due to experimental error.
For example, if extra drops of NaOH solution are inadvertantly added
from the buret, the volume recorded for NaOH solution added will be too high. Since we would divide by too large a
volume of NaOH solution in the final step, the calculated molarity of NaOH
solution would be erroneously low.
If
we know the volume and molarity of the acid in the Erlenmeyer flask as well as
the molarity of the metal hydroxide solution being added from the buret, we can
calculate the volume of metal hydroxide solution required for the titration, as
demonstrated in the following problem:
Sample
Exercise 10F:
How
many milliliters of 0.019 M strontium hydroxide are required to titrate 24 mL
of 0.026 M nitric acid?
Solution:

Chapter 10 Practice Exercises and Review Quizzes:
10-1) Write
a balanced equation for the combustion of each compound below using the
smallest possible whole-number coefficients:
(a) heptanol
(b) ethane
(c)
C4H4S
Click for Solution
10-1) (a)
2 C7H15OH + 21 O2 → 14 CO2 + 16 H2O
(b) 2
C2H6 + 7 O2 → 4 CO2 + 6 H2O
(c) C4H4S + 6 O2 → 4 CO2 + 2 H2O + SO2
10-2) Write a balanced equation for each reaction described below using the smallest possible whole-number coefficients:
(a)
barium metal reacts with oxygen gas
(b) rubidium metal reacts with water
(c) cesium oxide reacts with water
(d) calcium carbonate decomposes upon heating
Click for Solution
10-2)
(a) (product
is barium oxide = combine Ba2+ and O2- = BaO)
2 Ba + O2 → 2 BaO
(b)
2 Rb + 2 H2O → H2
+ 2 Rb+ + 2 OH-
(c) (cesium
oxide = combine Cs+ and O2- = Cs2O)
Cs2O
+ H2O → 2 Cs+
+ 2 OH-
(d) (calcium
carbonate = combine Ca2+ and CO32- = CaCO3,
CaCO3
→ CaO + CO2
10-3) What
is the molarity of each ion in the following
solutions?
(a) 0.18 M (NH4)2CO3
(b) 0.014 M FeBr3
Click for Solution
10-3) (a)
2 x 0.18 M = 0.36 M NH4+ and 1 x 0.18 M = 0.18 M CO32-
(b) 1
x 0.014 M = 0.014 M Fe3+ and 3 x 0.014 M = 0.042 M Br-
10-4) Indicate if any of the combinations
below yield no reaction and also write a balanced net ionic equation, including
states of matter, for any combinations that do yield a precipitate:
(i) aqueous lithium nitrate +
aqueous ammonium carbonate
(ii) aqueous sodium phosphate +
aqueous calcium acetate
(iii) Pb(NO3)2 (aq) + K2SO4 (aq)
(iv) NaI (aq) + NH4Br
(aq)
Click for Solution
10-4) No reaction will occur for (i) and (iv) as all combinations of cation +
anion will be soluble. One
precipitate will form in each of (ii) and (iii):
(i)
NH4+ (aq) + NO3- (aq) → no reaction
Li+ (aq) +
CO32- (aq) → no
reaction
(ii) Na+ (aq) + CH3COO- (aq) → no
reaction
net ionic equation: 3 Ca2+ (aq) + 2 PO43- (aq) → Ca3(PO4)2 (s)
(iii) K+ (aq) + NO3- (aq) → no reaction
net
ionic equation: Pb2+ (aq) + SO42- (aq) → PbSO4 (s)
(iv) NH4+ (aq) + I- (aq) → no reaction
Na+ (aq) + Br- (aq) → no
reaction
10-5) Write a
balanced molecular equation, including states of matter, for the neutralization
reaction between solutions of sulfuric acid and lithium hydroxide.
Click for Solution
10-5) H2SO4 (aq) + 2 LiOH (aq) → 2 H2O (l) + Li2SO4
(aq)
10-6)
If 13 mL of 0.24 M barium hydroxide solution is required to titrate 28 mL of
acetic acid solution, what is the molarity of the acetic acid solution?
Click for Solution
10-6)
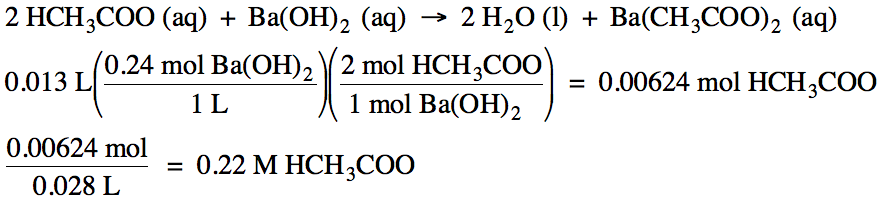
10-7)
How many milliliters of 0.0164 M hydrochloric acid can be titrated with 12.5 mL
of 0.0214 M calcium hydroxide?
Click for Solution
10-7)